
Interdependent Metal–Metal Bonding and Ligand Redox-Activity in a Series of Dinuclear Macrocyclic Complexes of Iron, Cobalt, and Nickel. Qiuran Wang, Shaoguang Zhang, Peng Cui, Alexander B. 
Cationic Phosphorus Compounds Based on a Bis(1-piperidinyl)-Substituted Carbodiphosphorane: Syntheses, Structures, and Csp3–H Activation. Alexander Kroll, Henning Steinert, Mike Jörges, Tim Steinke, Bert Mallick, Viktoria H.Journal of the American Chemical Society 2021, 143 Main Group Redox Catalysis of Organopnictogens: Vertical Periodic Trends and Emerging Opportunities in Group 15. Journal of the American Chemical Society 2022, 144 Versatile Fe–Sn Bonding Interactions in a Metallostannylene System: Multiple Bonding and C–H Bond Activation. Comparing Isoelectronic, Quadruple-Bonded Metalloporphyrin and Metallocorrole Dimers: Scalar-Relativistic DFT Calculations Predict a >1 eV Range for Ionization Potential and Electron Affinity. 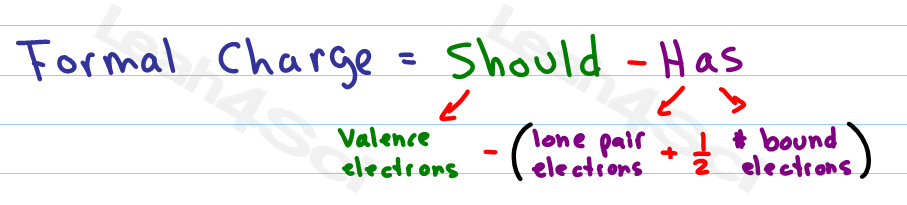
Jeanet Conradie, Hugo Vazquez-Lima, Abraham B.The Hyperporphyrin Concept: A Contemporary Perspective. Mechanistic Insights into the Oxidative and Reductive Quenching Cycles of Transition Metal Photoredox Catalysts through Effective Oxidation State Analysis. Edinson Medina, Christian Sandoval-Pauker, Pedro Salvador, Balazs Pinter.ACS Organic & Inorganic Au 2022, Article ASAP. Theoretical Photoelectron Spectroscopy of Low-Valent Carbon Species: A ∼6 eV Range of Ionization Potentials among Carbenes, Ylides, and Carbodiphosphoranes. Generally, the lowest energy structure is the one with the smallest formal charges on the atoms and the most distributed charge.This article is cited by 126 publications.Knowledge of the lowest energy structure helps in predicting the major product of a reaction and also describes a lot of phenomena.Formal charges help in the selection of the lowest energy structure from a number of possible Lewis structures for a given species.The formal charge being a theoretical charge doesn’t indicate any real charge rather separation in the molecule.A negative formal charge indicates that an atom is more likely to be the source of electrons while a positive one, on the other hand, it is more likely to take electrons, and most likely be the reaction’s site. 
The formal charge of a molecule can indicate how it will behave during a process. This condition could point to resonance structures, especially if the structures have the same atom arrangement but different types of arrangements of bonds. While formal charge can indicate a molecule’s preferred structure, the problem becomes more complicated when numerous equally preferred structures exist. The one with the least/lowest formal charges is the ideal structure. An atom in a molecule should have a formal charge of zero to have the lowest energy and hence the most stable state. Knowing the formal charge on a particular atom in a structure is an important part of keeping track of the electrons and is important for establishing and predicting the reactivity. Prior to discussing how to calculate formal charge(FC), we must understand that it is n essential, basic concept to master in order to better understand molecular structures and reactionsīasically, we can define formal charge as the hypothetical measure of charge assigned to an atom in a molecule, assuming that electrons in all chemical bonds are shared equally between atoms, regardless of relative electronegativity.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
